BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://ijmm.ir/article-1-1482-en.html

 , Shabnam Razavi2
, Shabnam Razavi2 

 , Malihe Talebi1
, Malihe Talebi1 

 , Faramarz Masjedian Jazi1
, Faramarz Masjedian Jazi1 

 , Abed Zahedi Bialvaei3
, Abed Zahedi Bialvaei3 

 , Maryam Mirshekar1
, Maryam Mirshekar1 

 , Vahid Lohrasbi1
, Vahid Lohrasbi1 


2- Department of Microbiology, Faculty of Medicine, Iran University of Medical Sciences, Tehran, Iran ,
3- Microbial Biotechnology Research Center, Iran University of Medical Sciences, Tehran, Iran
Klebsiella pneumoniae is an opportunistic Gram-negative bacterium associated with hospitals- and community-acquired infections. K. pneumoniae has evolved into two particular pathotypes: classical and hypervirulent K. pneumoniae (1). Classical K. pneumo-niae has been called a superbug dependent on its acquisition of different antimicrobial resistance genes and is basically responsible for hospital-acquired infections (2). Hypervirulent strains are more virulent than classical strains and are initially described to cause community-acquired infections (3).
The increasing global prevalence of antimicrobial-resistant bacteria, both in the hospital and in the community, jeopardizes the ability to effectively treat patients (4). Because these bacteria often show multidrug resistance (MDR) phenotypes, treatment options are severely limited (5). Therefore, it can be assumed that patients with serious infections will soon no longer be treatable with existing antimicrobial drugs. This resistance is mainly because of plasmids containing extended-spectrum β-lactamase (ESBL). ESBL enzymes in K. pneumoniae are mainly consisted of two types of enzymes SHV and TEM encoded by the plasmid, and were often associated with nosocomial outbreaks (6). Furthermore, there is an increasing rate of K. pneumoniae strains producing CTX-M type, and they are now considered to be the primary ESBL producers that are almost always associated with community-acquired infections (7, 8). Because the genes are simply spread via plasmid among the Enterobacteriaceae family, they can simplify the transmission of resistance to beta-lactams and other antibiotics (9). Moreover, the pathogenicity of K. pneumoniae is because of diverse virulence factors that empower the bacteria to conquer innate host immunity and to cause infection in a host (9, 10). K. pneumoniae virulence factors are lipopolysaccharides, capsule, the ability of biofilm formation, hyper-muco-viscosity, iron acquisition systems, serum resistance, and adhesins (11, 12).
K. pneumoniae with potency in acquiring antimi-crobial resistance genes and pathogenicity can be asymptomatically carried in the intestinal tract, nose, throat, and skin of healthy individuals. Studies of the gut flora of humans and animals give a more precise picture of the entire burden of antimicrobial resist-ance in society than data based on pathogenic iso-lates. As a matter of fact, the study of gut flora has shown that they may act as a reservoir for plasmids harboring antibiotic resistance genes, especially ESBL (13, 14). Moreover, drug resistance surveillance has reported that asymptomatic carriers in the population are mostly colonized with resistant bacteria, resulting in the infection (15, 16). So the study of gut flora is helpful in the understanding and monitoring of resistance mediated selection in the community (17). In addition, fecal carriage of antibiotic-resistant K. pneumoniae can be a community reservoir and lead to outbreaks. The current study was aimed to detect the antibiotic resistance pattern, the genetic determ-inants responsible for ESBL production (SHV, TEM, and CTX-M), carbapenem resistance (blaKPC), and other virulence genes [magA (Mucoviscosity associated gen), rmpA (hypermucoviscosity), fimH (fimbriae gene), BssS (biofilm formation)] in K. pneumoniae isolated from healthy volunteer’s feces.
Sample Collection and Questionnaire
The cross-sectional descriptive study was conducted among 350 fresh stool samples randomly selected from healthy volunteers (healthy volunteers to get a health card) submitted to the Northwestern Tehran Health Center between February and October 2018. A questionnaire was prepared for every volunteer who participated in the study to collect data regarding name, gender, age, medical history (History of hospi-talization or antacid and antibiotic intake), dietary habits (cheese, yogurt, milk, chicken, or meat consu-mption), and personal hygiene habits. People who had a history of using antibiotics, probiotics, and prebiotics and people with a history of chronic gastrointestinal infections were excluded from the research. The stool samples were cultured on MacConkey agar plates (Merck, Germany) and incubated at 37°C for 24 hours. The identifying process for K. pneumoniae isolates carried out using Gram staining and biochemical testing (methyl red, indole, citrate, vogus-prauskaur, urease, and mobility).
Susceptibility Testing
An antimicrobial susceptibility testing was done for each pure isolation by Kirby-Bauer disc diffusion method following the Clinical and Laboratory Stand-ard Institute guidelines (2018). The following antibio-tics were used: ceftriaxone (30 μg), piperacillin tazoba-ctam (100 μg), co-trimoxazole (1.25 μg), amikacin (30 μg), cefotaxime (30 μg), ciprofloxacin (5μg), ceftazi-dime (30 μg), cefipime (30 μg), meropenem (10 μg), imipenem (10 μg), and gentamicin (10 μg). Escherichia coli ATCC 25922 was used as negative controls (18).
MIC was performed to detect susceptibility to imip-enem, meropenem, ciprofloxacin and ceftriaxone using E-test strip (Liofilchem, Italy), following the man-ufacture’s protocol. Briefly, Mueller-Hinton agar plates (Merck, Germany) were inoculated with K. pne-umoniae strain suspensions adjusted to 0.5 McFarland standard turbidity and E-test strips were used. The MIC for each isolate was measured by reading the value at the point of intersection of the inhibition zone with the point on the scale of the E-test strip.
Analysis of Resistance and Virulence Genes using PCR Method
The extraction of bacterial DNA was performed by the boiling method (19). First, a colony of a bacterial isolate was added to 20 μL of tissue buffer consisting of 0.05 M NaoH + 0.25% SDS and incubated for 10 minutes at 95°C. After incubation, the mixture was centrifuged at 13,000 g for one minute, and 180 μL of Milli-Q water was added. The extracted DNA was frozen at -20°C for long-term storage.
The designed oligonucleotide sequences utilized to amplify fragments of the target genes are shown in Table 1. Polymerase Chain Reaction (PCR) was perfor-med in a total volume of 25 μL containing 12.5 µL of PCR Master Mix x2 (Amplicon, Denmark), 1 µL of each Forward and Reverse primer, 1 µL of DNA template, 9.5 µL sterile deionized distilled water, then DNA amplification was carried out with the thermocycler (Thermo Scientific, USA). Escherichia coli ATCC 25922, K. pneumoniae Strains ATCC 700603, and ATCC 7881 were used as controls.
Table 1. The sequences of primers used in this study
| Genes | Sequence 5ˊ to 3ˊ | Product size | Sources | Annealing temperature |
| magA | GCCAGTCCGAAAGTGAACGA | 162 | This study | 56 |
| TGCCAACAATTCCCGTTTCTG | ||||
| RmpA | TTCAGGGAAATGGGGAGGGT | 250 | This study | 60 |
| TTGCAGCACTGCTTGTTCCT | ||||
| fimH | ATCGTTAATCGCGGTGCTGA | 274 | This study | 54 |
| GGAGGCGGTATTGGTGAAGA | ||||
| BssS | AGCTACGATGCGCTGATGTT | 148 | This study | 58 |
| CAATCTTCGCAATGCCTGCT | ||||
| Blakpc | GCCGTCTAGTTCTGCTGTCT | 141 | This study | 60 |
| CGGTATCCATCGCGTACACA | ||||
| blaCTXM | CGCGTGATACCACTTCACCT | 77 | This study | 60 |
| GCTGTCGCCCAATGCTTTAC | ||||
| blaSHV | ACAGCTGGCTTTCGCTTAGT | 85 | This study | 60 |
| ATCTCCCTGTTAGCCACCCT | ||||
| blaTEM | CGTGTCGCCCTTATTCCCTT | 480 | This study | 60 |
| CTCCGGTTCCCAACGATCAA |
The PCR amplification consisted of an initial dena-turation at 95°C for 5 min, followed by 40 cycles of denaturation at 95°C for 40s, annealing for 40 s at temperatures specified for each primer, and extension at 72°C for 30s. This was followed by a final extension step at 72°C for 5 minutes. The PCR products were electrophoresed using 1% agarose gel and visualized by a gel documentation system.
The stool samples of 350 healthy individuals were collected from Northwestern Tehran Health Centers during a period of 8 months, from February 2018 to October 2018. The specimens were examined for microbial and biochemical tests in the laboratory of the Medical School of Iran University of Medical Sciences for the presence of K. pneumoniae. Of the 350-stool sample, all belonging to male individuals, only 60 isolates were identified as K. pneumoniae. In this study, the age range was 20-42 years, 67% were under 30. About 49% of these people also had nutrition-related occupations (Figure 1).
Among K. pneumoniae isolates, the highest resistance rate belonged to piperacillin-tazobactam (41.6%, n=25), meropenem (28.8%, n=17) and co-trimoxazole (18.3%, n=11), followed by cefotaxime (8.3%, n=5), ceftazidime, ceftriaxone (6.6%, n=4), cefepime (6.6%, n=4) and ciprofloxacin (1.6%, n=1), respectively. The MIC of meropenem-resistant isolates was above 4 (μg/mL), and for ceftriaxone, resistance was above 12 (μg/mL), the only ciprofloxacin-resistant isolate was above 8 (μg/mL). Figure 2 shows the antibiotic susceptibility of the isolates.
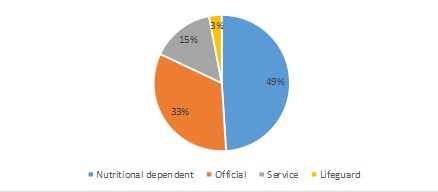
Figure 1. Occupations of the study subjects
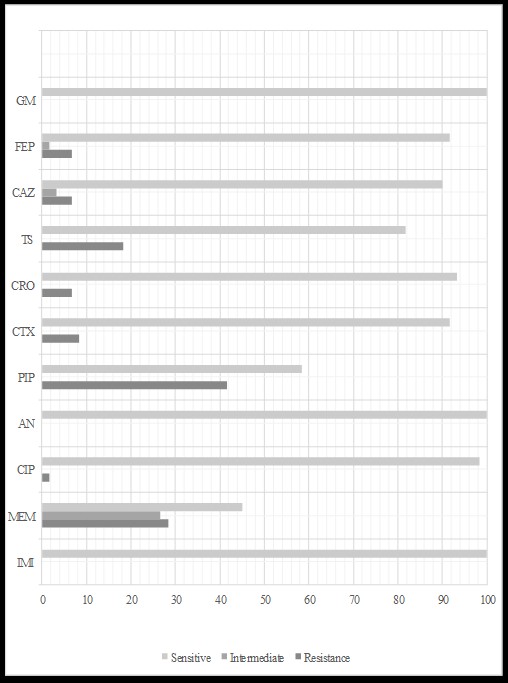
Figure 2. Percentage of antibiotic susceptibility in Klebsiella pneumoniae isolates
Among 60 isolates of K. pneumoniae, 95% (n=57) of isolates had fimH, 93.33% (n=56) had BssS, 45% (n=27) had rmpA, 41.66% (n=25) had blaTEM, 38.33% (n=23) had blaCTX-M, 35% (n=21) had blaSHV and 3% (n=2) had blaKPC, and none of these isolates carried magA gene (Figure 3). Out of 60 K. pneumoniae, 53 (88%) isolates had fimH and BssS genes simultaneously. MDR was observed in 4 (6.6%) cases of K. pneumoniae isolates. Out of MDR isolates, 3 had fimH and BssS genes.
Among all K. pneumoniae isolates, 28 (46.6%) had blaCTX-M, blaSHV, and blaTEM, (ESBLS) genes, of which 11 (18.3%) isolates belonged to patients in occupations related to the food industries. In addition, 21 (35%) ESBL positive isolates showed resistance to beta-lactams. Also, 8 (13.3%) isolates showed resist-ance to third-generation cephalosporins, of which 4 (6.6%) isolates had both blaTEM and blaSHV genes.

Figure 3. Gel electrophoresis results of virulence and antibiotic resistance genes - well 1: 100 bp DNA molecular marker, well 2: blaSHV, well 3: blaCTXM, well 4: blaKPC, well 5 BssS, well 6: rmpA, well 7: fimH, well 8: blaTEM genes
In this study, 60 K. pneumoniae isolates were identified in 350 stool samples obtained from comprehensive health examinations in Tehran, Iran. Our results confirm the high intestinal carriage of drug-resistant K. pneumoniae, in which the highest and lowest resistance rate belonged to piperacillin-tazobactam and ciprofloxacin, respectively. In addi-tion, the results showed that among all K. pneumoniae isolates, more than 90% carried fimH and BssS genes.
The reported asymptomatic carriage rates of ESBL positive Enterobacteriaceae in the feces of community dwellers vary in Asian countries: 5.0% in Japan (20), 40% in Thailand (21), and 50.6% in China (22). Most carriers were positive for CTX-M-type ESBL, which has nearly displaced other enzymes in Enterobacter-iaceae, including SHV and TEM variants. We showed that the prevalence of blaTEM, blaCTX-M, and blaSHV genes among the studied isolates were 35%, 38.33%, and 41.66%, respectively. However, Mansury et al. (2016) from Shiraz reported that the prevalence of blaTEM, blaCTX-M, and blaSHV genes among 38 ESBL producing K. pneumoniae were 16%, 19%, and 22.2%, respectively (23). In their study, the highest levels of resistance were related to amoxicillin (100%), cefotaxime (50%), and gentamicin (42.3%), and the lowest levels were related to meropenem (11.8%), amikacin and imipenem (both 15.9%). Asadpour et al., in 2016, determined the frequency of blaTEM and blaSHV genes in 34 broad-spectrum beta-lactamase producing K. pneumoniae by PCR (24). Of these, 40% of isolates carried blaTEM gene, and 23.07% had blaSHV gene. A Korean evaluation on stool samples of adults with comprehensive health examinations uncovered that from 109 fecal samples, only four blaSHV positive K. pneumoniae were identified (25). In another study carried out by Bing Lu et al., 22 K. pneumoniae isolates were detected from among 4340 stool samples collected from patients with diarrhea (26). Their results showed that all isolates were susceptible to ciprofloxacin, gentamicin, cefotaxime, ceftriaxone, nalidixic acid, imipenem, cefepime; the highest resistance rate belonged to amoxicillin-clavonate (100%) and ampicillin (72.7%) (26). The results showed that all strains were susceptible to imipenem, amikacin, and gentamicin, but little resistance was observed to ciprofloxacin, ceftriaxone and cefepime. In both studies, sensitivity to imipenem and gentamicin was observed, but cephalosporins may develop antibiotic resistance over time (8).
A study was performed by Candan and Aksöz (27) to evaluate the association among the genotypes of adhesins, mucoviscosity, capsules, carbapenem resis-tance, and other K. pneumoniae virulence factors. The OXA-48 and NDM-1 carbapenemase genes were detected in 33 and one from 50 K. pneumoniae strains, respectively. Also, KPC was not detected in K. pneumoniae isolates. Although no significant differe-nce was detected between susceptible and resistant strains because of the presence of virulence gene regions (P≥0.05), virulence factors were more diverse in carbapenem-resistant isolates (27). In research by Rodríguez-Navarro et al. (28), the genetic content of commensal and clinical strains was investigated. Their results indicated that the replicon profile was comparative in both populations, except for HI1 (only in commensal strains) and L, M, A/C, and N (detected only in clinical strains). Although F and I1 were the most common replicons, only IncI1, sequence type 12 (ST12) was related to blaCMY-2 in both populations. Also, the discoveries from this investigation affirm that healthy people strains have lower virulence factors and antibiotic resistance and show a higher diversity of phylogenetic lineages (in E. coli) than infection-causing strains (28).
Biofilm formation is one of the reasons for the increasing antibiotic resistance. fimH and BssS genes are involved in biofilm formation and ultimately may have a role in increasing antibiotic resistance (29). The frequency of fimH and rmpA genes in a study by El Fertas-Aissani et al. in Algeria were 100% and 3.7%, respectively (9). Another study was performed to evaluate fimH, and rmpA genes among K. pneumoniae isolates in hospitalized patients from Babol, Iran, which their frequency was reported 86.1% and 10.8%, respectively (30). Also, similar to our study, none of the isolates showed the magA gene in the two recent reports.
In summary, although the sample was collected from healthy individuals, some antibiotic resistance genes were observed. We were able to isolate K. pneumoniae from 350 stool samples from adults that underwent a medical examination. Since the treat-ment of infections caused by this bacterium is associated with many limitations, there is a need for effective measures to prevent and control the infection and careful use of antibiotics to control the rise and spread of fecal carriage of antibiotic-resistant pathogens.
None.
None.
Conflicts of Interest
There is no conflict of interest between the authors.
Received: 2021/09/12 | Accepted: 2021/11/10 | ePublished: 2021/12/8
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





