BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://ijmm.ir/article-1-1394-en.html


 , Asma Laktib1
, Asma Laktib1 

 , Mariem Zanzan2
, Mariem Zanzan2 

 , Rachida Mimouni3
, Rachida Mimouni3 

 , Fouad Achemchem2
, Fouad Achemchem2 
 , Aicha Aitalla4
, Aicha Aitalla4 

 , Fatima Hamadi5
, Fatima Hamadi5 


2- Bioprocess and Environment Team, LASIME laboratory, Agadir High School of Technology, Ibn Zohr University, Agadir, Morocco
3- Laboratory of Microbial Biotechnology and Plants Protection, Ibn Zohr University, Faculty of Sciences, Agadir-Morocco
4- Laboratory of Microbial Biotechnology and Plants Protection, Ibn Zohr University, Faculty of Sciences, Agadir-Morocco.
5- Laboratory of Microbial Biotechnology and Plants Protection, Department of biology, Faculty of Sciences, Ibn Zohr University, Agadir, Morocco ,
Acinetobacter is a genus of gram-negative bacteria belonging to the phylum of Proteobacteria. Its species are coccobacillus non-motile, non-fermentative, cata-lase and citrate positive, oxidase negative, growing under strictly aerobic conditions. Among these species, Acinetobacter baumannii (A. baumannii) is the most common nosocomial pathogen of hospital-acquired infections such as ventilator-associated pneumonia, bacteremia, urinary tract infections, burn wound infections, endocarditis, meningitis, and septi-cemia.
The Infectious Diseases Society has recently named Enterococcus faecium, Staphylococcus aureus, Klebsi-ella pneumoniae, A. baumannii, Pseudomonas aerug-inosa, and Enterobacter spp. as’the ESKAPE patho-gens. This is because of their resistance to antibiotics and their ability to escape from the antibiotic action.
In the last fifteen years, A. baumannii has received significant attention due to its several resistance mechanisms and high rates of morbidity and mort-ality. A. baumannii is naturally resistant to many antibiotics (1) and, due to the widespread use of antibiotics, the multidrug-resistant (MDR) and the extensively drug-resistant A. baumannii (XDR) have emerged worldwide (2, 3). Consequently, the treat-ment of diseases caused by this bacterium is impeded by this fact, limiting the therapeutic options (1).
Acinetobacter baumannii can adhere to several abiotic surfaces, including polystyrene, polypropylene, and borosili-cate. It was shown that A. baumannii colonizes various objects, including medical equipment and tools, hospital furniture, and even gowns and gloves of healthcare providers (4, 5). This pathogen can also adhere to biotic surfaces such as erythrocytes (6) and human bronchial epithelial cells (7).
In addition, the biofilm formation ability of A. baumannii is an essential factor contributing to the persistence in the hospital environment and the occ-urrence of nosocomial infections (8, 9). This capacity favors survival in hostile environments and acts as a barrier by reducing the activity of drugs and biocides (10).
Biofilms are bacterial clusters irreversibly attached to a surface or between cells, wrapped by a matrix composed of exopolysaccharides (EPS), proteins, extracellular DNA, and water channel (11). The production of EPS variously termed capsule, glycol-calyx, or a slime substance is a significant virulence factor (12). Bacterial adhesion, aggregation, and bacterial proliferation followed by slime production result in a biofilm capable of establishing a virulent and persistent infection on the implant (13). EPS leads to an irreversible adhesion that occurs with the formation of micro-colonies, the maturation of the biofilm (11), and the formation of three-dimensional biofilm structures (14). On the other hand, the extracellular matrix is composed of a variety of proteins with different functions and structures, including flagella, pili type IV, and amyloid-like pro-teins (e.g., curli protein in Escherichia coli (15) and FapC in Pseudomonas aeruginosa (16). Specifically, amyloid-like proteins have been recognized as one of the most important components involved in the assembly and integrity of the biofilm matrix (17). Besides, the physicochemical properties of the surface also have a rather important role in this phenomenon, namely the hydrophobicity and surface tension components (18).
The first aim of this work was to study the influence of the carbon source and temperature on exopolysaccharides and extracellular proteins produc-tion of Carbapenem-resistant A. baumannii strains. These strains were isolated from two hospital facility intensive care units (adult and neonatal intensive care units) (18). The second aim was to determine the ability of these isolates to form biofilm on silicone and ceramic surfaces and the effect of glucose on this latter. Ceramic and silicone surfaces are frequently found in hospital environments for that they were chosen for this work. In addition, the physicochemical properties of silicone and ceramic surfaces were determined. Finally, the correlation between EPS, extracellular proteins and, biofilm formation was examined.
Bacterial Strains
A total of 6 Carbapenem (imipenem and mero-penem) resistant A. baumannii (Ab) strains was studied. The strains were isolated from medical devices of two hospital intensive care units (adult and neonatal intensive care units) and identified by our laboratory (18). Also, the carbapenemase encoding genes were detected and identified (LAKTIB et al. under publication).
Screening for EPS Production
To determine EPS production, pure cultures were streaked on Trypticase Soy Agar (TSA) plates cont-aining 0.2, 0.8, 1, 1.5, or 2% of glucose or sucrose as carbon sources. They were then incubated at 25 or 37°C for 3 days. Strains that produced slimy colonies were recorded as capable of producing EPS (19).
Quantification of EPS and Proteins Production
-
Extracellular Matrix (ECM) Extraction
The strains of A. baumannii were incubated on TSA plates with or without glucose supplementation (0.2% and 1.5%) for 72 h at 37°C or 25°C. Subsequently, they were scraped off, suspended in 1 mL of 1.5 M NaCl solution, and incubated for 3 min at 25°C. The suspensions were then centrifuged at 5000 g for 10 min at 25°C, and the supernatants were collected as ECM fractions (20).
-
EPS Quantification
EPS was quantified by the phenol-sulphuric acid method with some modifications (21). In the test tube, 0.5mL of ECM sample was added to 0.5 mL of 5% phenol, followed by adding 2.5 mL of concentrated sulphuric acid. The test tube containing the solutions was then incubated for 10 min at 25°C, and optical density (OD) was measured at 492 nm. Glucose was used as a standard.
-
Protein Quantification
To quantify proteins in the ECM fractions, we used the Biuret assay (22). After adding 0.5 mL ECM, 0.2 mL Biuret reagent was added, and the mixture was then incubated for 30 min at 27°C. Subsequently, the absorbance was measured at 570 nm. Bovine serum albumin (BSA) was used as a standard.
Growth Conditions and Preparation of Microbial Suspension
After incubation at 37°C on TSA plates, the cells were harvested and washed twice by centrifugation for 15 min at 8400xg, then resuspended in physiologic water (NaCl: 9 g/L) to obtain a bacterial suspension of 108 CFU/mL.
Preparation of Substratum Surface
We used Silicone and Ceramic. These latter were cut into 1 cm2 square (10 _ 10 mm coupon-tests). Then 70% (vol/vol) ethanol solution was employed fo cleaning for 15 min and being then rinsed using distilled water. Finally, thesterilization of the substrates was performed by autoclaving for 20 min at 120°C.
Bacterial Adhesion
The incubation of 10 mL of bacterial suspension wa conducted in a Petri dish that contained substratum surface coupons (3 h at 37°C). After that, to remove the non-adhering bacteria, the coupons were rinsed 3 times using distilled water.
Biofilm Formation
The coupons with adhering bacteria were placed in a new Petri dish containing 20 mL of TSB with or without glucose (0.2% and 1.5%) and incubated for 72 h at 37°C. After that, they were washed three times with sterile distilled water and immersed in a test tube that contained physiological water (NaCl: 9 g/L). We detached the bacterial cells from the support utilizing a sonication bath (Power sonic 405) for 5 min. After sonication, CFUs were counted employing the serial dilution technique of the obtained bacterial susp-ension. The determination of the counts was done on nutrient agar plates after incubation for 24 h at 37°C by the plate count method. Each step was conducted 3 times.
Contact Angle Measurements and Hydrophobicity
Contact angle measurements were performed using a goniometer by the sessile drop method. One drop of a liquid was deposited onto a dry substratum surface, as it was done in he article by AZELMAD et al, 2018. Three to six contact angle measurements were made on each substratum surface for all probe liquids, including formamide (99%), diiodomethane (99%), and distilled water. The Lifshitz-Van der Waals (γLW), electron donor (γ−), and electron acceptor (γ+) components of the surface tension of the surfaces were estimated from the approach proposed by Oss et al. (1988). In this approach, the contact angles (θ) can be expressed as:
Cosθ= -1+2(γSLW γLLW)1/2/ γL + 2(γS+ γL-)1/2/ γL +2(γS– γL+)1/2/ γL
θ is measured by the contact angle. (S) and (L) denote surface and liquid respectively.
The cell surface hydrophobicity was evaluated through contact angle measurements and using the approach of Van Oss et al. (23), as it was done in he article by AZELMAD et al, 2018 . In this approach, the degree of hydrophobicity of a given material (i) is expressed as the free energy of interaction between two entities of that material when immersed in water (w) ΔGiwi. If the interaction between the two entities is stronger than the interaction of each entity with water ΔGiwi < 0, the material is considered hydrophobic. Conversely, if ΔGiwi > 0, the material is hydrophilic. ΔGiwi can be calculated through the surface tension components of the interacting entities, according to:
ΔGiwi= 2 γiw= -2[(γiLW)1/2 – (γwLW)1/2)2 + 2(γi+ γi-)1/2 + (γw+ γw-)1/2 – (γi+ γw-)1/2 – (γw+ γi-)1/2]
Statistical Analysis
Correlation analysis, one- and multi-ways ANOVA analysis of variance were applied using Statistica StatSoft version 6. The experiments were performed in triplicate. P-values of ≤ 0.05 were considered significant.
Screening for Eps Production
All tested strains of A. baumannii showed a different ability to produce slimy colonies by changing the added sugar, its concentration, and the incubation temperature (Table 1). This production was also affected by the type of strain. Ab6 appeared to be the most productive strain of mucoid colonies. A significant difference was observed between sucrose and glucose in both temperatures. Indeed, incubation at 37°C with glucose (1.5%) was optimal for maximum viscosity for all strains (Table 1) and, in particular, Ab6. On the other hand, at 37°C, viscosity raises by increasing the glucose concentration from 0.2 to 1.5% and decreases again by maintaining 2% (Table 1). On the contrary, there was no significant change by increasing the sucrose concentration, and the viscosity was very low compared to glucose (Table 1).
By incubating the strains at 25°C, the colonies were much less slimy than 37°C for glucose, and there was no significant difference between the concentrations (Table 1). Even when increasing the concentration, a complete absence of viscosity was observed for sucrose (Table 1).
Table 1. Formation of slimy colonies by A. baumannii strains in the presence of different concentrations (0.2, 0.8, 1, 1.5, or 2%) of sugars
| Glucose | Sucrose | |||||||||||||||||||
| 37°C | 25°C | 37°C | 25°C | |||||||||||||||||
| Strains | 0.2 | 0.8 | 1 | 1.5 | 2 | 0.2 | 0.8 | 1 | 1.5 | 2 | 0.2 | 0.8 | 1 | 1.5 | 2 | 0.2 | 0.8 | 1 | 1.5 | 2 |
| Ab1 | + | + | + | ++ | + | + | + | + | + | + | - | + | + | + | + | - | - | - | - | - |
| Ab2 | + | + | + | ++ | + | + | + | + | + | + | - | + | + | + | + | - | - | - | - | - |
| Ab3 | + | + | + | ++ | + | + | + | + | + | + | - | + | + | + | + | - | - | - | - | - |
| Ab4 | + | + | + | ++ | + | + | + | + | + | + | + | + | + | + | + | - | - | - | - | - |
| Ab5 | + | + | + | ++ | + | + | + | + | + | + | + | + | + | + | + | - | - | - | - | - |
| Ab6 | + | ++ | ++ | +++ | ++ | + | + | + | + | + | + | + | + | + | + | - | - | - | - | - |
Legend: -: no slimy, +: slimy, ++: high viscosity, +++: very high viscosity
EPS Quantification
EPS was quantified using the phenol-sulphuric acid method. At 37°C and in the absence of glucose, the EPS concentration ranged from 0.34 (Ab3) to 0.46 mg/mL (Ab6) (Figure 1a). At a concentration of 0.2%, the EPS produced by all strains decreased; for example, for Ab 6, the EPS production goes from 0.46 (without glucose) to 0.37 mg/mL (0.2% glucose). On the other hand, except for Ab3, the EPS production increased at 1.5% of glucose (Figure 1a). A significant difference between the concentrations was observed (P≤0.01).
At 25°C, the concentrations detected are very low compared to those found at 37°C (P≤0.0002). For both glucose concentrations, the EPS production decreased for all strains with significant differences (P≤0,04) (Figure 1b). In this case, it can be deduced that in addition to the stress caused by the temperature, glucose may have created another one, causing a decrease in EPS production.
To conclude, a glucose concentration of 1.5% and incubation at 37°C were the best conditions for optimal EPS production.
Protein Quantification
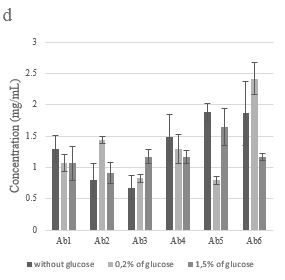
At 37°C, except Ab1, we observed that the proteins concentrations have increased for both glucose concentrations compared to its absence (Figure 1c). for the three strains (Ab2, Ab3, and Ab4), proteins concentration raised with the increase of glucose concentration. On the contrary, for Ab5 and Ab6, proteins production decreased when the glucose supplementation increased (Figure 1c). It appears that the maximum extracellular proteins concentration has been detected in the presence of glucose.
At 25°C, for all glucose concentrations, Figure 1d shows that adding glucose has caused an increase in proteins production for the three strains (Ab2, Ab3, and Ab6) and decreased this concentration for the others (Ab1, Ab4, and Ab5).
Biofilm Formation
All carbapenem-resistant A. baumannii strains have expressed their ability to form biofilm on ceramic and silicone surfaces but with different levels (Table 2).
Table 2. The number of A. baumannii adhered cells (104 CFU/cm2) forming a biofilm.
| Strains | Ceramic surface | Silicone surface |
| Ab 1 | 199.33 | 59 |
| Ab 2 | 63 | 483.33 |
| Ab 3 | 720 | 90 |
| Ab 4 | 37 | 45.67 |
| Ab 5 | 43 | 41 |
| Ab 6 | 64.33 | 65 |
This difference in biofilm-forming ability may be due to the physicochemical properties of the surfaces (Table 3).
Table 3. Contact angles (in degrees) of water (θw), formamide (θF), diiodomethane (θD), the surface tension of Lifshitz-Van der Waals (γLW), electron donor (γ–), electron acceptor (γ+) of silicone and ceramic surfaces and their free energy of interaction with water (ΔGiwi).
| Surface | Contact angles (°) | Surface tension: components and parameters (mJ.m–2) |
ΔGiwi (mJ.m–2) |
Hydrophobicity | |||||
| θD | θF | θw | ˠLW | ˠ+ | ˠ- | ||||
| Ceramic | 69,73 | 61,17 | 63,03 | 23,00 | 0,67 | 27,33 | 7,30 | Hydrophilic | |
| Silicone | 88,30 | 96,80 | 104,27 | 13,47 | 0,00 | 4,53 | -52,30 | Hydrophobic | |
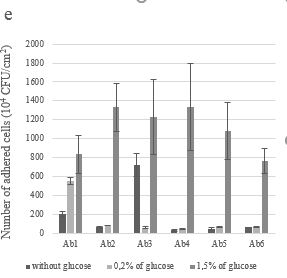
The effect of glucose on A. baumannii biofilm formation was also studied (Figure 1e, f). In the absence of glucose, the A. baumannii strains expre-ssed different preferences towards the two types of surfaces, taking the examples of Ab1, which adhered better to ceramic than silicone (P=0,001), and the opposite case of Ab2 (P=0,0002). By adding glucose, all strains formed a biofilm on silicone but to a lesser extent compared to ceramic except for Ab6, which formed more biofilm on the silicone with 0.2% of glucose than on ceramic. The greatest ability to form biofilm on the ceramic surface was expressed at a glucose concentration of 1.5% in all tested strains (P≤0.02). However, there was no significant difference in the majority of strains between the absence and 0,2% of glucose concentration (P≥0.06) except for Ab1(P=0.0004) and Ab3 (P=0.0009). For silicone surfaces, the addition of glucose did not significantly influence the biofilm format, except Ab2 and Ab6. We also observed that Ab6 reached its maximum at a glucose concentration of 0.2% (P=0.0002).
Biofilm Formation, Eps and Proteins Production
The correlation of the biofilm formation with the quantities of EPS and proteins produced at 37°C in different glucose concentrations was also studied. There is a positive correlation between EPS production and the numbers of bacterial cells forming a biofilm on the silicone with 0.2% of glucose concentration (R= 0.79).
For proteins, a positive correlation was established between the biofilm formed on the ceramic and 1.5% of glucose (R=0.85) (Figure 2a), knowing that the highest capacity of biofilm formation for the ceramic was found at this concentration (Figure 1). With a concentration of 0.2% glucose, a negative correlation was detected between protein production and biofilm formation on the silicone surface (R=-0.63) (Figure 2b) and a positive one on the ceramic surface (R=0.54) (Figure 2c). For the rest, no correlation was detected.
 |
 |
 |
 |
 |
 |
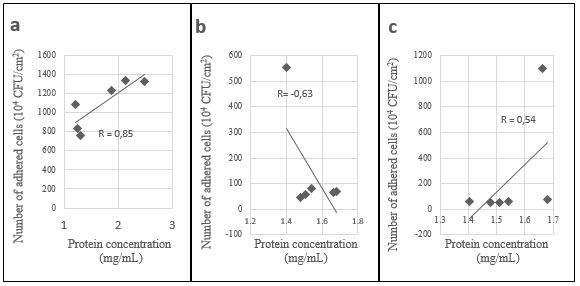
Figure 2. Correlation between proteins production and biofilm formation, on ceramic with 1.5% of glucose (a) and on silicone (b) and ceramic (c) with 0.2% of glucose concentration.
Our results have proved the powerful action of glucose on A. baumannii EPS and protein production and its biofilm-forming ability. Glucose can be both a carbon source and a signal molecule by affecting the expression of several genes and those involved in carbon utilization. In Pseudomonas aeruginosa, glucose availability is linked to the production of virulence factors (24). This may also be the case for A. baumannii following our results. In a study based on the connection between exogenous glucose and virulence in the opportunistic pathogen A. baumannii, 199 genes exhibited significantly differential expression levels when incubating this pathogen in glucose-supplemented Luria Bertani medium (25). 85% had higher transcription levels, while only 15% were down-regulated in this medium. In addition, the few genes down-regulated in the medium do not define any specific metabolism or biological processes while the other up-regulated are involved in cell surface composition, peptidoglycan, lipopolysacch-aride, EPS production, biosynthesis of EPS precursors, and secretion systems (25).
In most of the works available on databases, the EPS screening test based on slimy colonies was used for detecting lactic bacteria-produced EPS. Contradi-ctorily to our finding when glucose was the best for A. baumannii, a study showed that all strains of lactic bacteria were able to produce EPS only in the presence of sucrose, and no production was detected for other types of sugar (19). Sucrose was also optimal for EPS production by Leuconostoc lactis (26). Similarly, Jung et al. (2013) have also detected, by quantifying EPS production under various conditions, the highest levels of EPS in the presence of sucrose by Cronobacter sakazakii ATCC 12868 (27).
Besides the presence of glucose, the temperature change also influenced EPS production by the tested strains since 37°C was the most suitable. Similar to our work, in the study by Junkins and Doyle, wild-type Escherichia coli O157:H7 strain 932 produced the greatest amounts of EPS when grown at 37°C (28). On the other hand, EPS production by Lactobacillus rhamnosus strain C83 was 37% higher when it was incubated at 25°C instead of 37°C (100 mg/L compared with 63 mg/L) (29).
Comparing the EPS and proteins produced quantities, we can observe that proteins were more important than EPS (0.46mg/mL for EPS and 2.48-mg/mL for proteins as a maximum) even with changing culture conditions (Figure 1). This result is in line with the one described by another work which showed that proteins are the major component of the extracellular matrix of multidrug-resistant Pseudo-monas aeruginosa DC-17 isolated from dental caries, with 1.928 mg/mL to 162.3 mg/L only of EPS (30). Despite the crucial role of EPS, proteins have dominated the matrix more than EPS, which probably reflects the main function of these proteins. However, the structural information for many of these proteins is poorly understood. The amyloid-forming proteins (amyloid-like proteins) are one of the most important components of the extracellular matrix involved in the formation and stability of the biofilm (17).
Our results proved that these carbapenem-resistant A. baumannii strains could form biofilm on both ceramic and silicone surfaces. The ability of A. baumannii to form a biofilm on ceramic was also investigated by Ivanković et al. (2017) who marked the essential role of disinfectant/surface material interactions c in the survival of A. baumannii biofilms on this type of support. Moreover, ceramic surfaces may be an important source of infection in hospital environments because biofilm populations on ceramic were more resistant compared with those on glass (31). Silicone has excellent biocompatibility and biodurability properties, making it useful for implant-table biomedical devices. M'hamedi et al. (2014) have found that A. baumannii isolates could form biofilm on silicone (32). Another work compared biofilm forma-tion on polyvinyl chloride, silicone, and rubber latex endotracheal probes, have shown that A. baumannii had more susceptibility to rubber latex followed by polyvinyl chloride and silicone last (33).
Contact angle measurements suggested that ceramic is a hydrophilic surface while silicone is a hydrophobic one, which could affect biofilm forma-tion on these surfaces. It has already been proved that the physicochemical parameters of both surface and bacteria can largely affect the adhesion of bacteria and, subsequently, the formation of biofilm (34). An A. baumannii strain that is more hydrophobic has been shown to adhere better to silicone catheters and has also produced more biofilm (35). According to Azelmad et al. (2018), the adhesion of P. aeruginosa differs from one strain to another depending on the properties of the surfaces and of the strain itself (34).
Concerning the glucose effect on biofilm, the most remarkable ability to form biofilm on ceramic was detected at a glucose concentration of 1.5%. How-ever, for silicone surfaces, the addition of glucose did not greatly influence the biofilm formation. Generally, biofilm-producing bacteria are often associated with the increased synthesis of different compounds of ECM. ECM of biofilm received important attention from scientists in these recent years because of their crucial functions. In A. baumannii, increased prod-uction of ECM creates a protective environment that prevents antibiotic penetration and thus leading to resistance development.
Despite the intervention of EPS and proteins in the biofilm formation and the effect observed by the previous glucose results on the latter, the correlation analysis did not prove much. This lack of correlation may reflect some of the variations with this pathogen, which may pose a challenge in the treatment of biofilm formation. From these results, it can also be deduced that biofilm formation by these A. baumannii strains depends on several factors such as the strain itself, its intrinsic factors, the surface nature, and the surrounding environment.
In conclusion, the results showed that all clinical isolates of carbapenem-resistant A. baumannii tested were slimmer in the presence of glucose at 37°C. Glucose has also improved these strains' exopo-lysaccharides and proteins production and biofilm formation. A. baumannii can form biofilms on both silicone and ceramic surfaces. The results obtained in this paper confirmed that biofilm is a multifactorial character. It involves different factors like bacteria, its determinants, and the capacity of these bacteria to produce EPS and proteins. In addition, the growing conditions (i.e., glucose amount and temperature) and surface properties of both cell surface and substratum also have a significant effect on this phenomenon. Depending on these factors, Carbapenem-resistant A. baumannii probably uses different approaches to form a biofilm, increase its hospital colonization, and increase its morbidity and mortality. Therefore, we must understand these approaches more to select an appropriate anti-A. baumannii biofilm therapy. Further work is necessary to elucidate the effects of growth conditions on biofilm formation, EPS, and pro-teins production and discover the different strategies used by this bacterium to persist and infect in the hospital environment. On the other hand, the environment and its components must be considered when studying the formation of biofilm, especially when it concerns pathogens because a simple change can alter the bacterium's behavior.
None.
There is no funding; the research was supported by our university.
Conflicts of Interest
We declare that there are no conflicts of interest.
Received: 2021/07/20 | Accepted: 2022/01/26 | ePublished: 2022/02/10
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |





