BibTeX | RIS | EndNote | Medlars | ProCite | Reference Manager | RefWorks
Send citation to:
URL: http://ijmm.ir/article-1-1285-en.html
2- Department of Cell and Molecular Biology and Microbiology, Faculty of Biological Science and Technology, University of Isfahan, Isfahan, Iran ,
Hydroxyapatite (HA) is one of the most important bioceramics utilized in medicine, and it has been intensively explored in recent years. It has a chemical composition and structure that is remarkably similar to bone and teeth minerals. After being implanted in the body, this bioactive and biocompatible substance interacts well with bone tissue and forms a direct bond with it (1, 2). HA binds, grows, and stimulates the synthesis of new bone tissue and stimulates bone growth into its porous space. This compound has the bone tendency, doesn't absorb and decompose, and ultimately achieves good adhesion to bone tissue (3, 2). Therefore, due to the compatibility of HA with bone tissue and soft tissues, this substance is the best substitute for hard tissues of the body such as bones and teeth. Generally, HA forms up 70% of human hard tissue as a mineral phase. The term "apatite" refers to a group of compounds that are structurally similar but not chemically identical. As a result, the term apatite refers to a description rather than a substance (4, 2).
The current study is a review, and all sources were gathered and retrieved from authentic databases in English. The search was conducted using the keywords nano, hydroxyapatite, biological synthesis, nanobiote-chnology, and microorganisms on Google Scholar, Science Direct, and PubMed. It should also be noted that this research is based on articles published by the corresponding authors, Dr. Giti Emtiazi and Dr. Rasoul Roghnian.
1-Properties and crystallography of HA
The apatite family is crystallized by the arrangement of a hexagonal prism with parallelograms. HA also ideally has a hexagonal structure with dimensions of 2 x 30 x 50 nm and has a specific composition of Ca10 (HPO4)6 (OH)2 and a definite crystallographic str-ucture. The chemical composition of pure hydroxya-patite is as follows: 39.68 percent calcium, 18.45 percent phosphorus, with a weight ratio of calcium to phosphorus of 2.151 and a molar ratio of calcium to phosphorus of 1.67. (5).
HA has a complex and relatively large single cell in which the integrity is likely to be lost. When the atomic ratio of calcium to phosphorus in hydroxyapatite is 1.6, it is totally stoichiometric. Therefore, due to the possibility of changes in the structure and its comp-osition, which leads to the formation of nonstoichio-metric HAs. Phosphate ions, calcium ions, and hydroxyl groups in HA can be replaced by other ions. These changes have a special effect on physical pro-perties, crystal structure, the molar ratio of calcium to phosphorus, and the solubility of HA under physiolo-gical conditions (1, 2). Although it is insoluble in water, this substance is regarded as a degradable material (with slow disintegration rate) (6).
2-Applications of HA
The main applications of HA in medicine, dentistry, diagnostics, catalysis, and sewage treatment are briefly outlined here. Direct application in orthopae-dics (7), bone tissue engineering (8), implant coating (9), antibacterial effect (10), drug delivery systems (11, 12), and application in dentistry (15) are some medical applications.
In general, bone tissue can self-repair; however, in cases such as serious bone tissue damage and aging, the bone’s ability to self-repair is eliminated. The researchers then introduced HA for replacement in bone tissue. Because of HA's biocompatibility and bioactivity, it can drive the growth of bone cells into its porous space, resulting in bone cell differentiation and proliferation. Therefore, this material is used in bone tissue engineering and bone cement enginee-ring. Therefore, HA has made significant progress in orthopedics and the placement of implants and bone grafts. HA is also used in oral and maxillofacial imp-lants to increase the height of decayed gums that are unable to mount dentures and implant grafts. Tooth enamel caries can also be partially regenerated using HA toothpaste, according to studies. Because HA has a high affinity for biological substances, it can lead to successful transplantation by replacing drugs, especia-lly antibiotics and immunosuppressive drugs, in im-plants coated with HA (7-12, 15).
Other applications such as non-medical included bacterial concentration (13), removal of radioactive metals (14), use in biosensors (16), and the use of HA as a catalyst (17).
3-Methods of synthesis of HA
Although calcium phosphate compounds had a wide range of uses in recent years, the optimal production methods are still being researched, particularly in the case of HA. Numerous chemical and biological meth-ods are used to synthesize this substance, as follows (1).
3-1- Chemical synthesis of HA
Nowadays, various chemical methods are used to produce HA nanocrystals, which depending on the type of method, produce products with different structures, appearance, and crystalline characteri-stics. Generally, chemical methods for the synthesis of HA can be divided into two categories: wet chemical synthesis methods and dry chemical synthesis methods (18). Dry synthesis methods or anhydrous mechanical synthesis methods include solid-state reaction and mechano-chemical reaction (19).
Considering raw materials, wet chemical reactions, including acid-base method, sol-gel method, ultrasonic-chemical synthesis, microwave synthesis reaction, multiple emulsion technique using temp-lates or micelles, chemical deposition technique, pre-paration method using conversion hydrothermal and soluble ignition method. These methods usually use soluble salts such as calcium nitrate and calcium chlo-ride, ammonium dihydrogen phosphate or diammon-ium hydrogen phosphate, and potassium phosphate. Table 1 summarizes the chemical synthesis methods of HA. (1, 19).
Table 1. Comparison of chemical synthesis methods of HA (1, 18, 19).
| Crystallinity | Morphology | Size | Conditions and mechanisms of synthesis | Precursors | Method | |
| Almost high | variable | More than 500nm | Temperatures above c1000 | CaCO3 CaHPO4.2H2O |
Solid-state | Dry synthesis |
| Almost high | Variable | More than 200nm | Mill with speed above 600rmp | CaO CaHPO4 |
Mechano-chemical | |
| Low | Variable | Micron | Reaction between acid and base | Ca (OH)2 H3PO4 |
Acid-base | Wet synthesis |
| Almost high | Generally spherical | Nano | Molecular mixing of calcium and phosphorus in a jelly-like atmosphere | Urea EDTA Ca (NO3)2 NH2PO4 |
Sol-ge | |
| Almost low | Generally needles like | Nano and micron | Create bubbles, burst bubbles and collide raw materials | Ca (OH)2 H3PO4 Ca (CH3CO)2 PO(OCH3)3 |
Ultrasonic-chemical | |
| Almost low | Variable | Micron | Heat and radiation waves | Ca PO4 |
Microwave | |
| Almost low | Generally spherical | Nano and micron | Dissolve the reactant in water and emulsify water in oil | Ca PO4 |
Multiple emulsion | |
| Low | Variable | Micron | Sedimentation | Ca (NO3)2 (NH4)3HPO4 |
Chemical deposition | |
| Almost low | Variable (Generally dandelion like) | Micron | High pressure, high temperature | Ca PO4 |
Conversion hydrothermal | |
| Almost low | Variable (Generally needles like) | Nano and micron | Add oxidizing agents | NH2PO4 Ca (NO3)2 |
Soluble ignition |
According to the findings of Hsieh et al., the sol-gel method has advantages over the other chemical methods listed in Table 1. This method requires low-temperature treatment compared to other methods. Because chemical methods usually use very high temperatures to form crystals. However, the temp-erature required for the HA formation operation in the sol-gel method is much lower. It is more cost-effective than others, and compared to the products of other methods, the HA synthesized from this method has a unique form. It is also possible to achieve nano size in this method. Another advantage of sol-gel compared to other chemical methods is the ability of this method to synthesize HA nanoparticles with almost higher purity (40).
3-2- Biological synthesis of HA
The bone tissue of most mammals, poultry, and fish is a major source of HA. This type of HA, as opposed to its synthetic type, is called biological or natural HA. Other sources of natural HA include coral, shrimp shells, snakeskin, hedgehog thorns, and some plants and algae. One of the most common sources of natural HA is mammalian bones such as livestock. This compound is obtained during long heat treatment at temperatures of about 1000 degrees Celsius. One of the major advantages of natural HA over chemical HA is that its physical structure and chemical makeup are identical to that of human bone tissue. For bioce-ramics like HA, the presence of a porous struc-ture with appropriate porosity is highly desirable, and the natural kind of HA provides this porous structure. The chemical composition also greatly affects the perfor-mance of HA. Large crystal bridges and large amounts of calcium oxide result in lower biocompatibility of HA. Components from the decomposition of HA also cause pH changes around the planting site. The benefits of natural HA include the presence of trace elements in bone tissue. Elements such as magnesium, sodium, potassium, iron, zinc, and other similar elements are present in very small amounts in bone tissue and play an important role in the process of bone repair and re-generation. Another significant advantage of natural HA is its relatively inexpensive and cost-effective process (20, 21).
3-2-1- Bacterial Synthesis of HA
One of the new methods of synthesis of nanosized and natural HA is the microbial method or biominera-lization method by microorganisms. As Emtiazi et al. previously proved that the bacterium can produce copper oxide and zinc oxide nanoparticles and listed many properties such as antibacterial properties for them, we decided that microorganism should be stud-ied as a factory for the production of other nanopar-ticles (22-26).
3-2-1-1- The role of Phosphatase Enzyme in the Formation of HA
Biomineralization is the process through which HA settles in the extracellular matrix during the osteoge-nesis process. Physiological mineralization occurs in hard tissue while calcification occurs in soft tissue. Tissue non-specific alkaline phosphatase (TNAP) hyd-rolyzes pyrophosphate and supplies inorganic phosph-ate to promote mineralization. This enzyme is also present in the bacterium and the process mentioned in this microbe is also possible (28).
Generally, there are two types of phosphatase enzy-mes in microorganisms. Generally, Acid phosphatase is outside the bacterial membrane, and Alkaline phos-phatase is located in the periplasmic region of the bacterial membrane (27, 29). Acid phosphatase enzy-mes can be used to make HA, although nanometer-sized HA crystals are larger since these enzymes are extracellular. Furthermore, the optimal pH for the activity of acidic phosphatase enzymes is below 5.8, at which point HA is demineralized and eliminated, albeit with the use of bacteria in the pericardial region that contains alkaline phosphatase. However, by using bacteria that have a high level of alkaline phosphatase in the periplasmic region and controlling the condi-tions, nanometer dimensions of HA with bioco-mpatibility and bioactivity properties can be achieved. The optimum pH for the activity of alkaline phosph-atase enzymes is 9 (4, 30).
All alkaline phosphatase enzymes belong to the hydrolase enzymes group, which nonspecifically tran-sfer phosphate groups from all phosphomonoester compounds [R-O-PO3]n to hydroxyl-containing polya-mines (OH) such as ethanolamines. In the structure of these metalloenzymes, there are two protein monomeric chains, two active zinc nuclei, and two cofactor sites for magnesium and calcium (Figure 1).
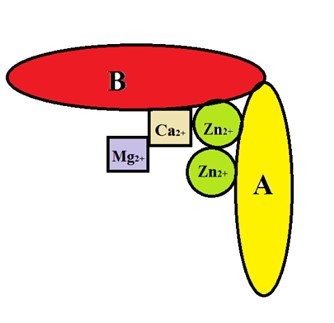
Figure 1. Schematic picture of alkaline phosphatase metal-oenzyme. A and B: protein monomeric chain, Zn2+: active site of zinc, Mg2+ and Ca2+: cofactor site for magnesium and calcium. (Source: Authors)
The function of these enzymes can be described as follows: first, they release phosphate groups from phosphomonoester compounds. Then offer phos-phate groups to hydroxyl in the limited periplasmic space. In the next step, cation attachment of Zn, Mg, and Ca will complete the nano crystallin HA synthesis.
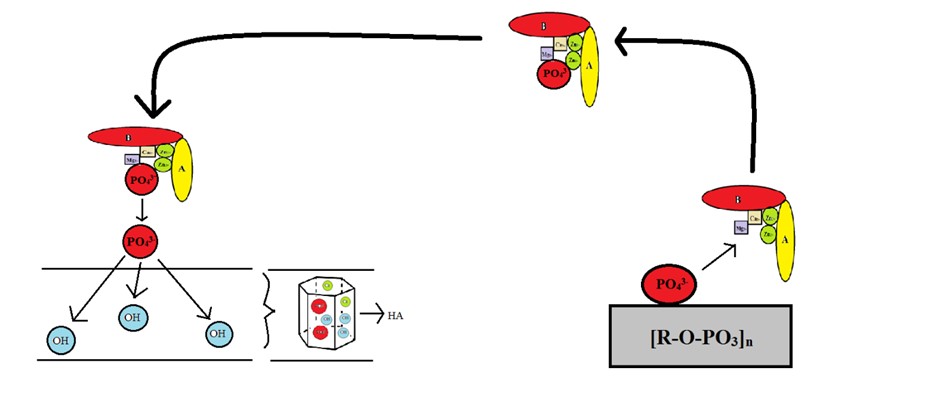
Figure 2. Expected process of HA crystals formation in bacteria and the role of alkaline phosphatase enzyme in this process. (Source: Authors)
Some gram-negative bacteria contain a group of alkaline phosphatase enzymes that can be hydro-xylated by using these bacteria and adding simple phosphomonoester compounds such as glycerol-2-phosphate, using an alcohol receptor such as Tris, and by adding calcium ions, synthesized nanometer apa-tite in the bacterial periplasmic space. This spatial constraint plays a key role in the formation of HA in the form of hexagonal prisms and nanometer crystal clusters (32).
3-2-1-2- The role of urease enzyme in the formation of HA
The enzyme urease is widely found in bacteria and is often used to produce microbial calcite. There are four regulatory models for the synthesis of urease enzymes in the microbial system. Constitutive urease is expressed steadily and independent of external conditions in the cell. Inducible urease is induced by an inducing molecule such as urea or other enviro-nmental conditions. Repressible urease is suppressed in the presence of ammonia, which is removed under nitrogen restriction and enzymatic activity is increa-sed. Finally, developmental urease means that the organism expresses a variable expression of urease in different evolutionary stages (33, 34).
Most microorganisms, with their ability to break down urea, use it as a source of nitrogen that is transported into the cellular cytoplasm by active transfer or inactive diffusion, where hydrolysis of urea releases ammonia molecules. Ammonia can then be absorbed directly by the glutamine synthetase-gluta-mate synthase pathway or by the action of glutamate dehydrogenase. An ideal microorganism for bio-cementation should be resistant to high conce-ntrations of urea and calcium. This microorganism often has to have a high level of urease activity that is permanently produced or can be significantly induced. Urease-producing bacteria are divided into two groups based on their response to ammonia. There is a group in which the enzyme activity is not suppressed in the presence of ammonia and a group in which the urease activity is suppressed (34, 35).
In the synthesis of urease HA, different amounts of CaCl2 and NaH2PO4 are used, in which case the decom-position of urea by urease causes homogeneous depo-sition of HA, due to the release of OH ions during the process of hydrolysis of urea. The carbonate ions formed in these reactions can also enter the crystal structure of HA particles. Thus, a type of carbonate HA is formed that may have a very similar structure to apatite in human bones. The experimental results show that reducing the ratio of material concentration to urease concentration reduces the particle size to less than 100 nm and their morphology changes to a spherical shape. One of the advantages of urease enzyme in the formation of HA is that the presence of urease allows the decomposition of urea at a lower temperature and the enzyme is not consumed during the reaction (36).
3-2-2- Fungal synthesis of HA
In the results of HA experiments, it has been observed that some fungi such as Aspergillus niger have the ability to stimulate the synthesis of HA in Potato Dextrose Agar (PDA) medium with appropriate concentrations of substances such as Na₂HPO₄ and CaCo₃, which are the main cause of HA crystals formation. Fungal metabolism produces an acidic subst-ance to dissolve CaCo, and the growth of micelles causes the uptake of Ca2+, which leads to calcium-rich levels to promote the production of secondary apatite, which is eventually converted to HA (Figure 3) (37).
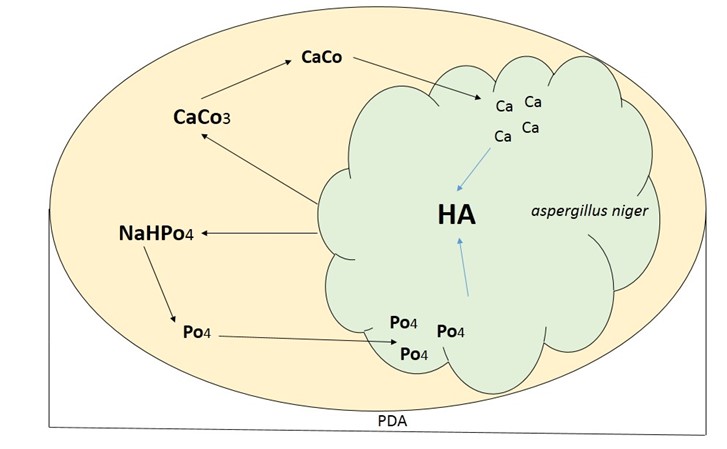
Figure 3. Predicted trend for HA formation in micelles and fungal hyphae (Source: Authors)
Alternatively, Aspergillus niger and wheat bran (as a source of phosphorus) have been used for the synthesis of HA. Wheat bran contains a significant amount of phosphorus (in the form of phytic acid), which is considered a cheap agricultural waste. This reaction is performed by using an extracellular phytase enzyme produced by fermentation of wheat bran with the mentioned fungus. Phytase is a class of phosphohydrolases that catalyzes the hydrolysis of phytic acid to the mineral monophosphate. Finally, calcium chloride is added to the fermented mushroom and wheat bran mixture, which a white precipitate is observed in the mixture after 24 h. Following the identification procedures, HA with characteristics similar to human bone HA was discovered. Nanos-tructured HA is naturally the major component of bone and teeth in humans.
HA has been produced for bone tissue engineering, implant coating, bone cement engineering, drug delivery systems, and other medical and non-medical uses in recent decades using different chemical and biological methods. Various chemical approaches have been proposed for the production of HA up to this point. According to the results and studies, the sol-gel approach is more suitable than other methods in the chemical synthesis of HA (1, 39 - 41). Generally, chemical methods have disadvantages compared to biological and natural approaches. These disadvantages included the need for precise pH adjustment, the lack of biocompatibility, the high cost of raw materials, and finally that it is not possible to achieve high purity of crystallinity and uniformity in shape and size. On the other hand, microbial HA size is between 25 and 30 nanometers (which is very close to natural HA in the human body). This subject draws the attention of scientists to the synthesis of microbial HA. The size and shape of the produced particles are homogeneous and have appropriate crystallinity in microbial synthesis, which improves the material's biocompatibility and bioactivity while also contri-buting to the mechanical qualities of the final HA part. Production of microbial HA is one-step, inexpensive, non-toxic, and has high purity. Unlike chemical synt-hesis, it does not require temperature treatment and precise pH adjustment and is easily achievable. Also, the antibacterial properties of these particles are very significant. However, with these interpretations, microbial synthesis of HA on a large scale is not possi-ble as much as chemical synthesis. However, because bone cell differentiation does not necessitate a large amount of HA, this kind of HA can be used in cell differentiation (1, 27, 42, 43).
Microbial HA in animal in-vivo experiments has shown the ability of osteogeneration and degrad-ability than chemically synthesized samples. Due to this property, in addition to cost-effectiveness, it is possible to achieve more suitable HA for medical purposes by developing a microbial approach.
Bio-nano-HA can play an important role as a revol-ution in nanomedicine. Bone injuries in modern life and the risk of rejection of bone implants have led scientists to discover new materials to improve bone repair, and scientists have turned to life sciences to build alternatives to bone grafts. Due to the morpho-logical similarity of biological HA with natural HA and its dimensional similarity (at the nanoscale), signi-ficant application of this material is expected in the nanomedical industry, bone tissue engineering, and bone cement engineering. The use of engineering principles in the creation of tools to study, modify, and produce tissue from natural or synthetic sources is known as bone tissue engineering. The primary goal of this approach is tissue regeneration and organ func-tion improvement through the use of three-dimensi-onal HA scaffolds. Cells from the patient's body (or human or animal transplant cells) or bone marrow are implanted into these scaffolds, which are then implan-ted in the patient's body. These scaffolds need to be as similar as possible to bone tissue. The engineering of bone cements is also created as a filler for cavities and bone defects, the most important of which is nano-HA (7). Biological nano-HA is used to make com-posites, dental filling cement, and oral and maxilla-ofacial implants. It was offered as a substance to prevent the formation of dental enamel decay and its partial restoration in the form of nano-HA toothpaste. There have also been reports of this bacterial bio cement used in the laboratory to repair rabbit jaw bone (15, 4).
None.
Conflicts of Interest
The authors did not report any conflict of interest.
Received: 2021/03/2 | Accepted: 2021/07/11 | ePublished: 2021/08/16
| Rights and permissions | |
 |
This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License. |








